Cane toad evolution
One of the most surprising results from TEAM BUFO’s studies has been the discovery of how incredibly fast cane toads have evolved during their Australian invasion. Understanding this rapid evolution in toads is central to developing ways to control them.

TEAM BUFO applies evolutionary concepts to the study of cane toads. We hope Charles Darwin would have approved.
Image by Melanie Elphick.
We normally think of “evolution” as a very slow process, taking
thousands or even millions of years. Evolution is the process by which
living creatures change gradually over time. It was suggested by
Charles Darwin, who also came up with a brilliant idea about the reason
that living beings change in this way. His idea - "natural selection" -
is based on the simple fact that characteristics that help animals to
survive or reproduce will tend to become more and more common (because by definition, the animals or plants with those characteristics will survive and
reproduce) whereas characteristics that make a creature less likely to
survive or reproduce will die out. So long as those characteristics can
be inherited from parent to offspring, evolution will inevitably occur.
That is, the frequencies of different genes will change through time,
and so will the traits of a species.
For more information on Evolution in Biology, read this Wikipedia article.
A short biography on Charles Darwin.
More information about Charles Darwin and his Theory of Evolution.
So if evolution is usually so slow, how can toads have evolved within the 70 or so years since they reached Australia?
Part of the reason lies in the advantages of very rapid dispersal in a species that is expanding its range. At the invasion front, there are very few other toads so the vanguard toads don’t have to compete with lots of other toads for access to food. It’s Toad Paradise. As a result, natural selection has favoured toads that travel very quickly. Because those toads can stay in the front of the invasion, they get more food and so can have more babies. It’s straightforward natural selection for speed.
But there’s a second reason why the toads have
accelerated - and the
answer lies in a very special KIND of evolution. It’s a very new idea,
and our research on Aussie cane toads has provided the first
really detailed evidence to support it. We’ve been able to publish our
results
in the very biggest scientific
journals.
It’s exciting to see Australian cane toads appearing in textbooks of evolutionary biology!
University of Melbourne’s Dr. Ben Phillips (a
foundation member of TEAM BUFO) is a leading expert in this emerging field of
research. Ben studies how invasions affect evolutionary processes. So he builds
mathematical models, as well as measuring toads. Ben studies how the invasion
process affects evolution. As toads spread across the Australian tropics, they
inevitably began to sort themselves out in terms of how quickly they
were moving. Slow toads ended up well behind the invasion front,
whereas quick toads were the only ones at the front. Basically, if you
are too slow you get left behind.

In the course of their Aussie travels, toads have evolved to be much quicker invaders
This simple process means that at the end of each generation, the
animals at the invasion front are the fastest dispersers - the toads
that have moved every night, all night, whenever conditions allow it. These athletic
toads breed with each other (it’s called the “Olympic Village Effect”)
and so produce fast-moving babies. And only the very fastest of those
babies will be the ones at the invasion front next year. And so on,
and so on ... Every generation, this process ends up increasing the
average speed of toads at the front. The end result was that the toads
that were first to arrive at Fogg Dam were really big (bigger toads are
quicker), had longer legs compared to their bodies (longer-legged toads
are quicker), and were amazingly active (moving every night, often more
than a kilometre every night) and very directional in their movements
(they all headed northwest). In comparison, cane toads radio-tracked in
Queensland, earlier in the Aussie invasion (by Professor Ross Alford
and Dr Lin Schwarzkopf, our colleagues at James Cook University) were
nomadic, meandering around rather than racing as fast as they could in a
straight line. As a result, the Queensland toads moved a bit less than
10 metres per day on average, whereas the Fogg Dam toads averaged about
ten times that much. An incredible shift in behaviour in less than 70
years!
As a result of that evolutionary shift in toads, the invasion front has
been moving faster and faster – from about 10 km per year in the early
days (1960’s) up to around 40 to 60 km per year now.
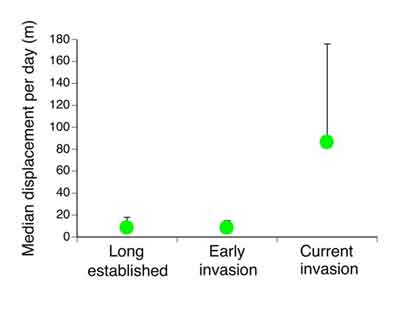
This graph shows how far cane toads move each day, comparing slow Queensland toads to faster NT ones.
Image by Melanie Elphick.
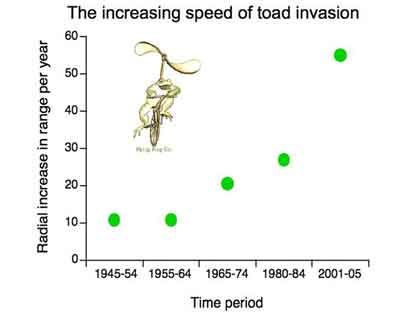
This graph shows how fast cane toads have travelled across tropical Australia. Image by Melanie Elphick.
That incredible increase in toad speed has put enormous pressure on the toads’ bodies – bodies that had evolved to sit around a swamp in Brazil and eat flies, not sprint across Australia like an Olympic long-distance athlete.
TEAM BUFO's veterinary pathologist, Dr Cathy Shilton, discovered that many of the
toads at the invasion front at Fogg Dam – especially the bigger, older,
longer-legged animals – have severe spinal arthritis. When Dr Greg
Brown tested the hopping ability of these toads (and same-sized animals
with normal spines), he found that the toads with arthritis had to take
shorter hops, and were obviously highly stressed and probably in a lot
of pain. But when he radio-tracked them, they moved just as far as
“normal” toads … cane toads really are tough!
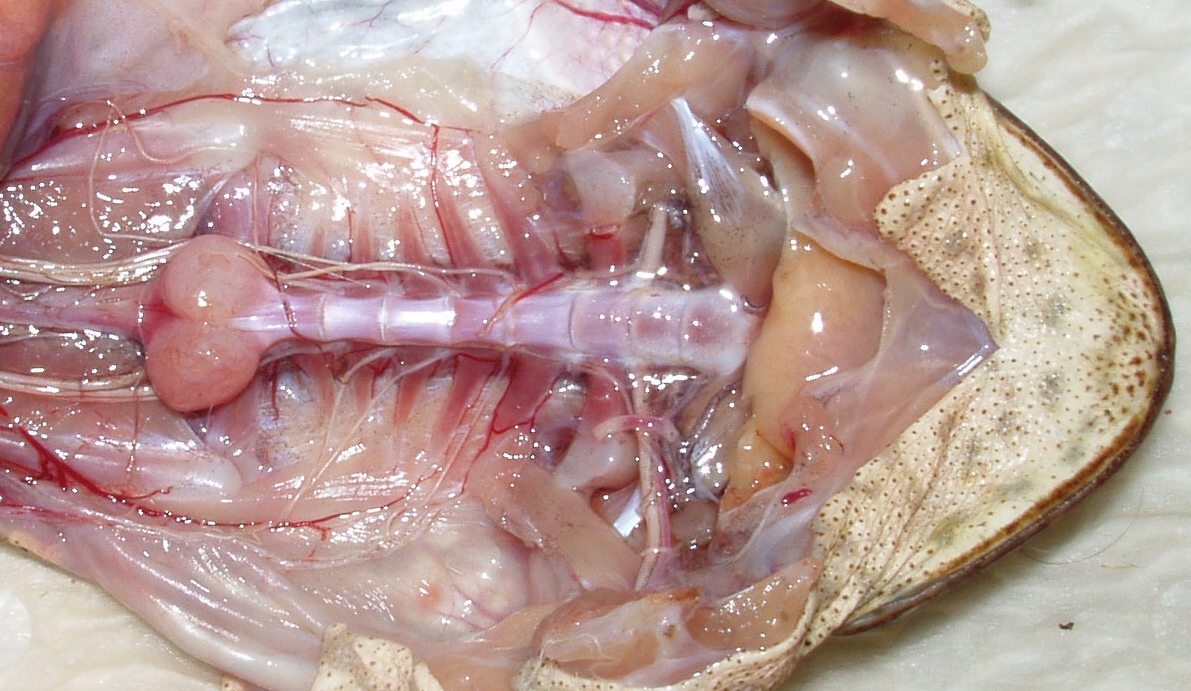
An arthritic cane toad spine. The huge lump at the rear of the spine is a massively arthritic joint. Photo by Cathy Shilton.
Is it really evolution?
TEAM BUFO student Cam Hudson measured thousands of cane toads, and the results are clearcut. Toads at the invasion front (in Western Australia) are very different in body shape to toads at the core of the species’ range in Australia (that is, in Queensland). The Queenslanders are short and stout, like the original toads in French Guiana, and the toads in Hawai’i. But after they were brought to Australia and released on the northeast coast, toads changed as they spread westwards.
As we mentioned in another page, those changes extend all the way to behaviour (invasion-front toads are very bold and active) and to physiology (invasion-front toads are better at travelling long distances without needing a rest).
But is this really evolution? To check that interpretation, we need to know WHY toads from different areas look and act differently. For example, it could just be due to toads experiencing different conditions as they grow up – perhaps something special about Western Australian ponds makes toads develop with long legs and active personalities?
We have two kinds of data to test this idea.
First, we have now sequenced the genome of the cane toad – that is, we know the genetic building-blocks that combine to make a toad. And sure enough, there are many differences in genes between toads in eastern Australia and toads in Western Australia.
Second, we have shown that those characteristics are inherited from parent to offspring. We did this by collecting toads from a range of areas across Australia, breeding them at our research stataion, and raising the offspring in captivity. When they grew large enough to radio-track, we released them and followed them. The invasion-front toads took off for the far horizons just like their parents did, the generation before) whereas the Queenslanders sat around. And the offspring also resembled their parents in body shapes and in other distinctive features like their immune systems. They even inherited the boldness of their parents. So now we have indisputable evidence that cane toads have evolved rapidly in Australia, to become better dispersers.
Return to Home Page
Toad genetics
How have cane toads managed to evolve so rapidly in Australia? In less than a human lifetime, toads have changed from being squat sit-at-home amphibians to become active, lithe long-distance runners. Well, perhaps not “lithe”. But they have certainly moved in that direction. That shift is especially puzzling because of the very low number of toads originally brought to Australia in 1935. There were only 101 original colonists, so much of the genetic variation present in the native range of the species (in South America) was left behind. Evolutionary change can only occur if it has the raw materials it needs – variation in traits – so how did this small number of pioneer toads give rise to descendants with major evolutionary modifications?
To answer that question, TEAM BUFO collaborated with geneticists from several institutions to sequence the genome of the cane toad. Assembling the blueprint to build a species (that is, documenting all of its genes, and working out their functions) used to be a massive challenge, taking many decades and many millions of dollars. But methods have improved, so we managed to assemble the genome very quickly and very cheaply. It was published in 2018.
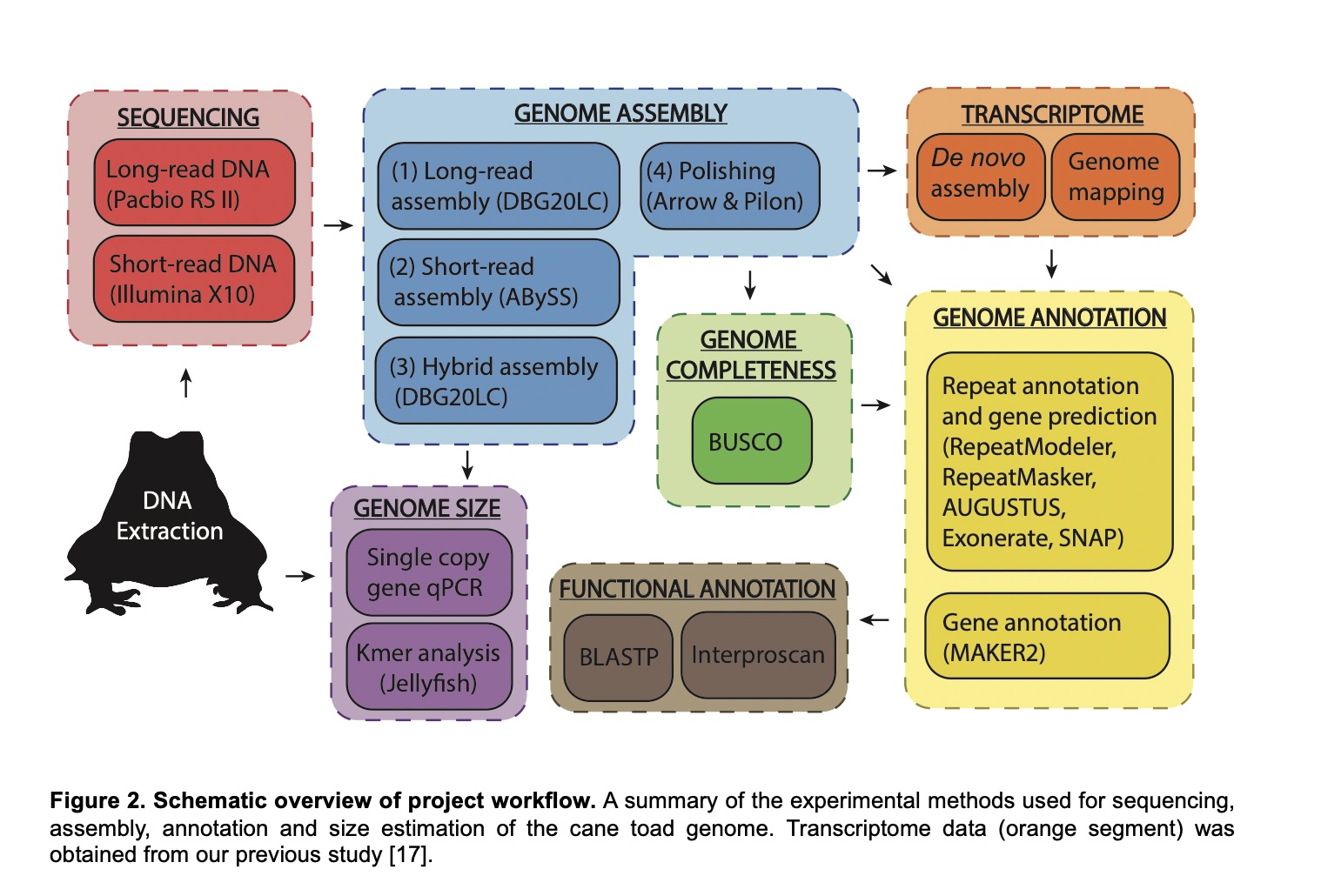
Sequencing the genome of an organism like the cane toad is a highly technical task. You need to work out how many genes there are, how they are arranged on chromosomes, and what functions each gene performs. This diagram explains the methods for people who has expertise in this field. Rick doesn't understand it.
With that background information, we can now ask specific questions about evolutionary changes that have occurred as the toads have spread across Australia. That work is already yielding exciting results. For example, we have confirmed that toads in different parts of Australia have very different patterns of gene expression. But strangely, their overall genetic constitution is not too divergent. We suspect that evolution has achieved these changes not by changing the array of genes within the toad’s genome, but by switching existing genes on or off. This mechanism is known as “epigenetics”, and it can provide a very rapid way for an organism to modify its traits.
Using evolution to control cane toads
At first sight, our discoveries about rapid evolution in cane toads don’t seem to be relevant to toad control. But in fact, evolutionary thinking can be very useful in dealing with a biological invasion.
As we’ve explained, cane toads at the invasion front in tropical Western Australia are very different from the cane toads that were first brought to Australia (and to the ones that still live in Queensland, where toads have been present for more than 80 years). The invasion-front animals are the offspring of invasion-front parents, so for 80 years the animals at the vanguard of the invasion have been evolving to be different from the range-core animals. For example, at the invasion-front there are very few other toads, so there is less competition for food. Evolution has favoured traits (like long legs) that enable toads to disperse more rapidly, rather than to compete with other toads.
Because those changes have a genetic basis, we can ask a novel question: what would happen if the two types of toad (invasion-front and range-core) came into contact? We expect that the range-core animals would outcompete the invasion-front ones both for food and for mates, because evolutionary pressures for success in those tasks has been stronger at the range-core than the invasion-front for 80 years. So, what would happen? The greater success of animals carrying range-core genes (which make toads focus on competition not dispersal) would tend to break up the new trait combinations that have evolved at the invasion front. As a result, the offspring from such a mixed population would behave more like range-core toads than invasion-front toads – which could be very good news for managers who want to slow down the invasion.
This kind of “genetic backburn” would work best if it occurred just before the toads reached a wide barrier of unsuitable (seasonally dry) habitat. Invasion-front toads might be able to cross even a wide barrier, because they can travel up to 60 km during the wet-season. But range-core toads have no hope of making it across within a single wet-season – and so, would be killed by dry conditions long before they could reach the other side.
A long stretch of dry country south of Broome provides just this kind of opportunity. The only water for most of the year comes from artificial stock-watering points, and we could make this inaccessible to toads (because these animals are poor climbers). If we did that, and introduced range-core (Queensland) toads at the same time, we might be able to stop the toad invasion in its tracks. If it worked, this plan could stop toads reaching the Pilbara region further south, where conditions would be suitable for them.
Of course, the plan might not work. Cyclonic rains might create a wet corridor that lasted for many months, enabling even slow toads to make it to the other side. But the costs of trying are low, and the potential benefits are huge. Feasibility studies by Reid Tingley and Ben Phillips are encouraging. If the “arid corridor” idea works, it will be a spectacular example of how basic research can suggest novel solutions to difficult environmental problems. And how we might be able to turn the toad’s evolution against them.

The country south of Broome, in northwestern Australia, is highly arid. The desert meets the sea, leaving little suitable habitat for toads to disperse south towards the Pilbara region, where conditions would be more favourable for them. So perhaps if we turned off artificial watering places, or made them inaccessible to toads, we could prevent the toads moving south? The plan might work best if combined with a "genetic backburn" - that is, we release stay-at-home Queensland toads just north of the arid barrier, to breed with the dispersing toads and so create a less mobile animal. Photo by Terri Shine.

For the full story about the cane toad invasion of Australia, and what we can do about it, read Rick’s book “Cane Toad Wars” (published by University of California Press, 2018). The book is written for the general public, not for scientists, and is aimed at a wide audience. The book can be purchased through online bookstores like Amazon, or you can buy a copy through your local bookstore.
Return to Home Page
